 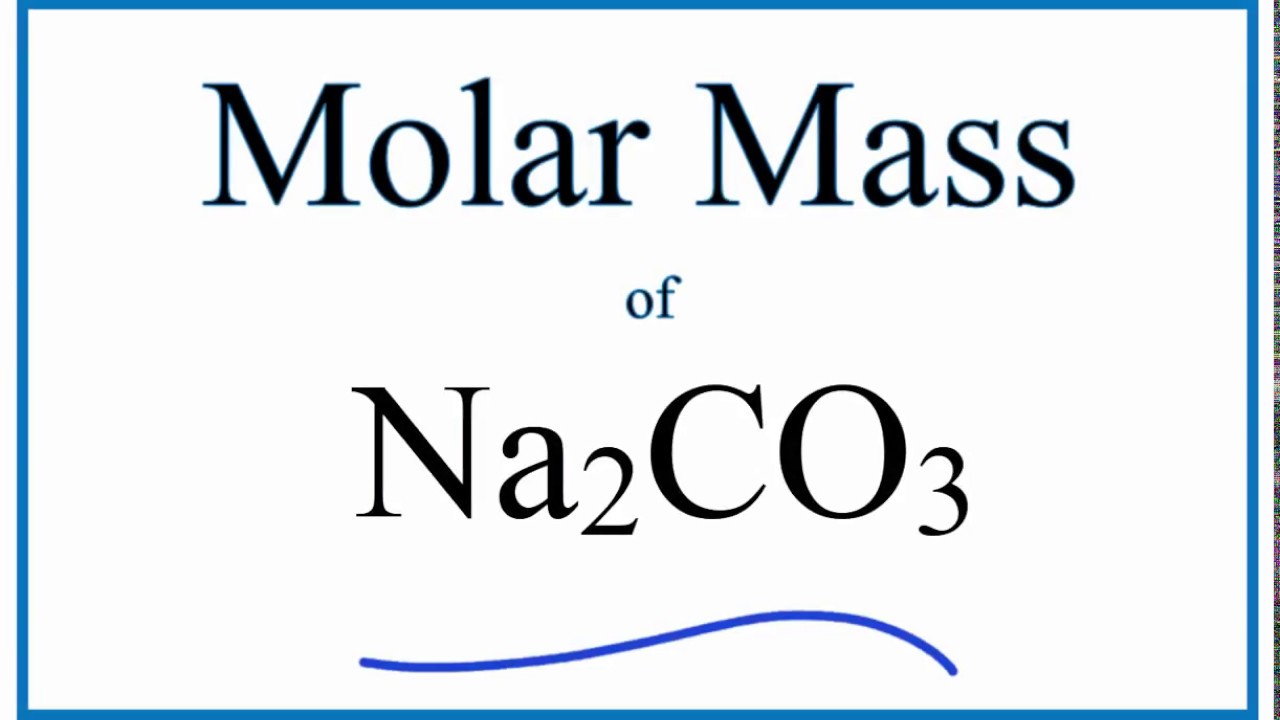
On the basis of above data, the average atomic mass of carbon will be. Of course then I start to wonder how molar weights of other elements are defined, with varying isotopic combinations, and I have the same question. Isotope Relative abundance() Atomic mass(amu) 12 C 98.892 12 13 C 1.108 13.00335 14. I tried googling for "standard carbon isotope ratio" but didn't find anything definitive. I dont think your formula is correct, but heres a general strategy for this type of problem: Divide the mass in grams by the molar mass of the compound and. There are two major isotopes of Carbon C - 12 and C - 13 found in nature, having an. With the ratio of each, the average value comes out to 12.0107.īut where does that number come from? Is it by definition? Is it by convention? Is it a practical number? Where do the error bars come from? For instance, I would expect to see different isotope distributions at different geographic locations (or what about the moon, asteroids, Jupiter, etc.). Calculating the atomic mass of Carbon: The atomic number of Carbon is 6. 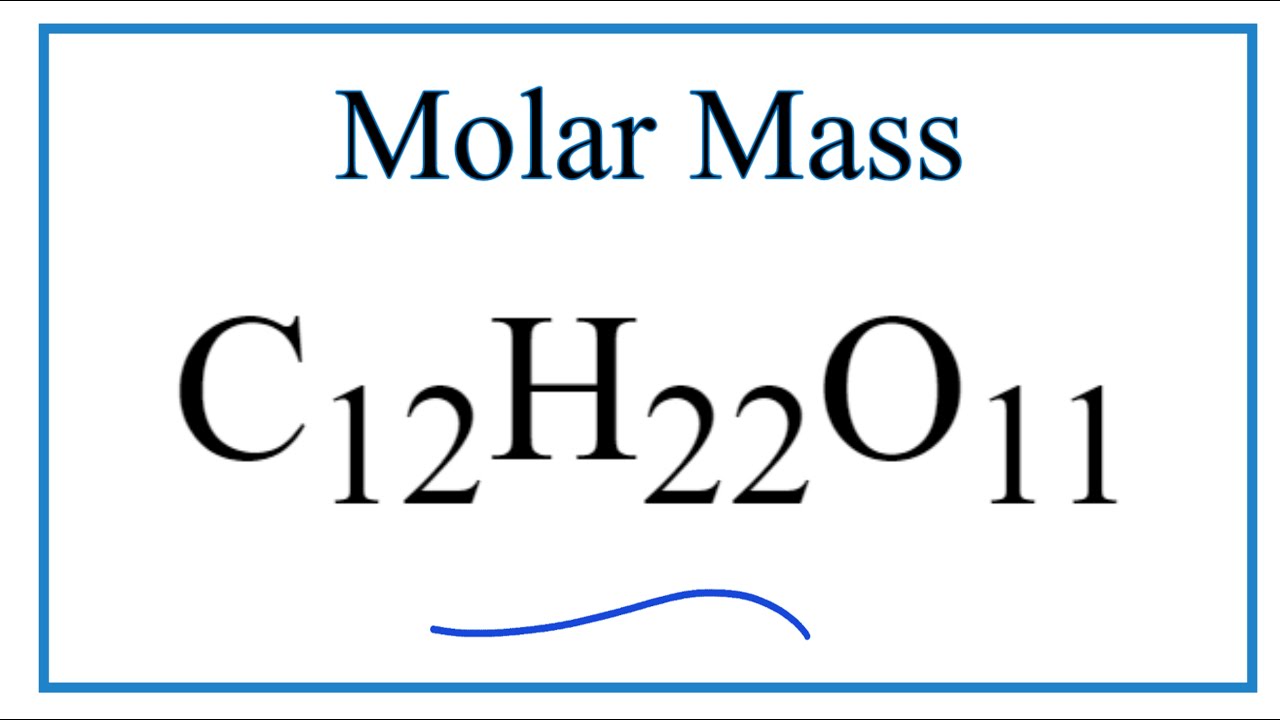
Place the following steps for calculating relative atomic mass in the correct. Percentages can be entered as decimals or percentages (i.e. Enter an optional molar mass to find the molecular formula. The most stable artificial radioisotope is 11 C, which has a half-life of 20.3402 (53) min. This is also the only carbon radioisotope found in nature, as trace quantities are formed cosmogenically by the reaction 14 N + n 14 C + 1 H. To calculate the empirical formula, enter the composition (e.g. The longest-lived radioisotope is 14 C, with a half-life of 5.70 (3) × 103 years. When I asked on a forum why this isn't precisely 12, I was told that typically carbon is a mixture of isotopes which have relative atomic masses of 12, 13 and 14. Why is the mass in amu of a carbon-12 atom reported as 12.011 in the. Instructions This program determines both empirical and molecular formulas. However, when I go to websites such as or and I ask what is the molar weight of Carbon, I get answers such as: So how good is our atomic radius? Standard tables give the atomic radius of strontium is in the range 192-220 pm.The mole is defined as the amount of a chemical substance which contains as many representative particles, e.g., atoms, molecules, ions, electrons, or photons, as there are atoms in 12 grams of carbon-12 (12C), the isotope of carbon with relative atomic mass 12 (from Wikipedia). molar mass of carbon-12: Numerical value: 11.9 x 10-3 kg mol-1: Standard uncertainty: 0.0 x 10-3 kg mol-1: Relative standard uncertainty: 3. Be able to find the number of atoms or molecules in a given weight of a substance. 
Define molecular weight, formula weight, and molar mass explain how the latter differs from the first two. If you had a simple compound like methane, CH4, approximately 1 in every 100 of these molecules will contain. Molar mass unit (g/mol) Molar mass is a macroscopic (large scale) unit of masses of atoms or molecules. Be able to calculate the number of moles in a given mass of a substance, or the mass corresponding to a given number of moles. Carbon-13 makes up 1.11 of all carbon atoms. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12. Since 3 3=27 and 4 3 = 64, you know that the cube root of 55 will be between 3 and 4, so the cube root should be a bit less than 4 × 10 –8. The value of 1 a.m.u is 1.66 x 10 -24 g which is exactly the mass of 1 mole of carbon atoms. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
